The 20th Conference of the Asian Pacific Association for the Study of the Liver
Poster Presentation(PP230)
Hepatology International.2010, 4(1):157
Bang-Fu Wu1,2, Jiang-Ying Yang2, Fang-Qin Li1, Yun Zhou2, Wen-Bao Zhu 1, Fu-Xin Lin1, Yan-Ping Fu1, Xue-Song Li1, Wei Zheng1, Wei Chen1,
Jun Yang2, Hui-Hua Zhou1, Chun-Qiong Hou1
1Gastroenterology and Hepatology Center, Tongji Medical College Affiliated Dongguan Hospital, Huazhong University of Science and Technology, Liaobu Southwest Road, No.171-175, Dongguan, China,
2 Guangzhou Pubang Biological Immune Technology Research Center, Room 904, D District, Guangzhou International Business Incubator, Guangzhou Science City, Luogang District, Guangzhou, China
Background: To observe the treating effects of HBsAg pulsed autologous dendritic cells (anti-HBV-DC) derived from peripheral blood mononuclear cells (PBMC) combine thymosin-a1 for the HBeAg negative chronic hepatitis B (CHB) patients.
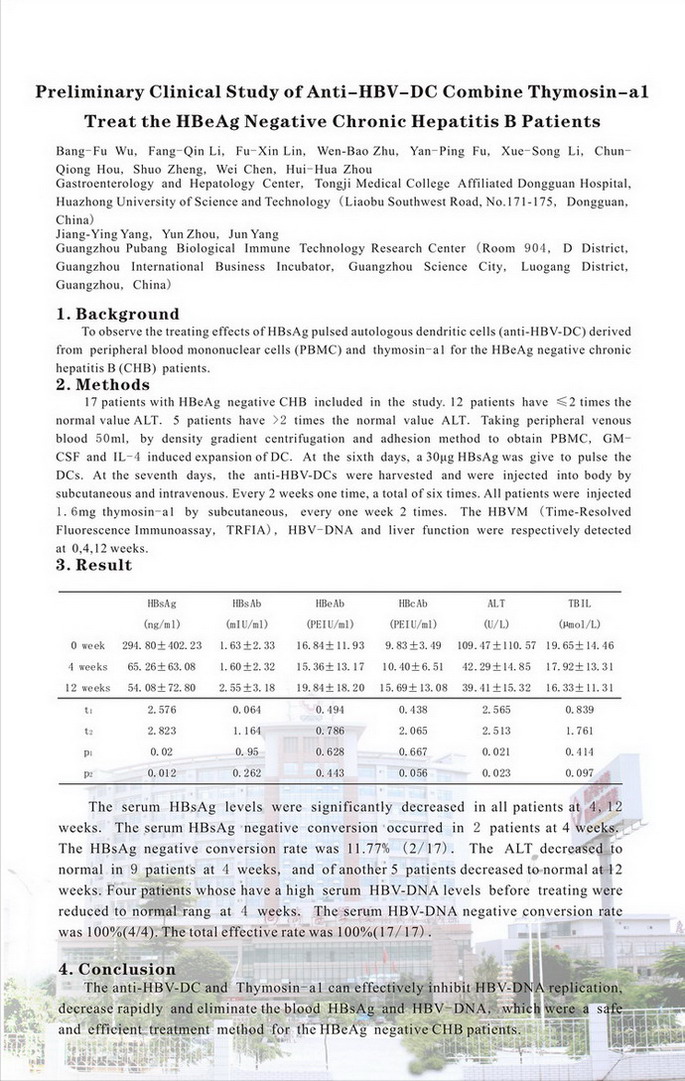
Methods: 17 patients with HBeAg negative CHB included in the study. 12 patients have ≤2 times the normal value ALT. 5 patients have>2 times the normal value ALT. Taking peripheral venous blood 50 ml, by density gradient centrifugation and adhesion method to obtain PBMC, GM-CSF and IL-4 induced expansion of DC. At the sixth days, a 30 μg HBsAg was give to pulse the DCs. At the seventh days, the anti-HBV-DCs were harvested and were injected into body by subcutaneous and intravenous. Every 2 weeks one time, a total of six times. All patients were injected 1.6 mg thymosin-a1 by subcutaneous, every one week 2 times. The HBVM (TRFIA), HBVDNA and ALT were respectively detected at 0, 4, 12 weeks.
Result: At 0, 4, 12 weeks, the serum HBsAg levels were (294.80 ± 402.23) ng/ml, (65.26 ± 63.08) ng/ml(t = 2.58, p = 0.02), and (54.08 ±72.80) ng/ml(t = 2.82, p = 0.012); the serum ALT levels were (109.47 ± 110.57) U/L, (42.29 ± 14.85) U/L(t = 2.57, p = 0.021), and (39.41 ±15.32) U/L(t = 2.51, p = 0.023). The serum HBsAg levels were significantly decreased in all patients at 4, 12 weeks. The serum HBsAg negative conversion occurred in 2 patients at 4 weeks. The HBsAg negative conversion rate was 11.77% (2/17). The ALT decreased to normal in 9 patients at 4 weeks, and of another 5 patients decreased to normal at 12 weeks. Four patients whose have a high serum HBVDNA levels before treating were reduced to normal range at 4 weeks. The serum HBVDNA negative conversion rate was 100% (4/4). The total effective rate was 100% (17/17).
Conclusion: The anti-HBV-DC combine Thymosin-a1 can effectively inhibit HBVDNA replication, decrease rapidly and eliminate the blood HBsAg and HBVDNA, which were a safe and efficient treatment method for the HBeAg negative CHB patients.
抗HBV-DC联合胸腺肽a1治疗e抗原阴性慢性乙型肝炎的初步临床研究
第20届亚太肝脏研究会年会(北京)
海报展示(PP230)
Hepatology International.2010, 4(1):157
华中科技大学同济医学院附属东莞医院消化肝病中心 吴邦富 李芳琴 凌佛鑫 朱文宝 付彦平 李雪松 侯春琼 郑硕 陈伟 周慧华
广州普邦生物免疫技术研究院 杨江英 周赟 杨军
目的 观察HBsAg致敏自体外周血单个核细胞(PBMC)来源的树突状细胞(抗HBV-DC)联合胸腺肽a1治疗HBeAg阴性慢性乙型肝炎(CHB)的临床效果。
方法 HBeAg阴性CHB患者17人接受临床研究。ALT≤2倍正常值者12例,ALT>2倍者5例。取肝素抗凝外周静脉血50ml,以密度梯度离心及贴壁法获得PBMC,GM-CSF和IL-4诱导扩增DC,第6天给予30μg的HBsAg致敏DC,第7天收获抗HBV-DC,皮下和静脉各注射1/2。每2周1次,共6次。皮下注射胸腺肽a1每次1.6mg,每周2次。分别于0、4、12周检测HBVM定量(时间分辨荧光免疫分析技术,TRFIA)、HBV-DNA定量及肝功能。
结果 0、4、12周的HBsAg分别为(294.80±402.23)ng/ml、(65.26±63.08)ng/ml(t=2.58,P=0.02)和(54.08±72.80)ng/ml(t=2.82,P=0.012);ALT(109.47±110.57)U/L、(42.29±14.85)U/L(t=2.57,P=0.021)和(39.41±15.32)U/L(t=2.51,P=0.023)。全部患者治疗后4、12周的HBsAg均明显下降其中2例的HBsAg 4周时转阴,转阴率11.77%(2/17)。9例ALT 4周时降至正常,另5例ALT 12周时降至正常。治疗前HBV-DNA定量升高者4例,治疗后4周均降至正常范围,HBV-DNA转阴率100%(4/4)。总有效率100%(17/17)。
结论 抗HBV-DC联合胸腺肽a1治疗,可有效地抑制HBeAg阴性CHB患者的HBV-DNA复制,快速降低并清除血中HBsAg和HBV-DNA,部分患者获得治愈,是一种安全、高效的治疗HBeAg阴性CHB患者的方法。
|


